What Are they?An easy way to remember them is by using the acronym OIL RIG. Oxidation and Reduction are used to describe changes in substances in reactions. To help us understand what oxidation and reduction actually are, we'll use the example of Copper ions becoming copper. Here, the copper is gaining electrons, so it is being reduced. This is a reduction reaction. If this reaction was reversed, it would show the ionisation of copper. In this reaction, Oxygen is losing electrons so it is being oxidised. Usually in this equation, you'd move the electrons to the right hand side but I'm keeping them on the left to help you visualise what's happening What's Redox?A Redox reaction is a reaction where oxidation and reduction are happening at the same time. For example, the reaction of Copper with Oxygen, the Oxygen loses 2 electrons (gets oxidised), and the copper gains 2 electrons (gets reduced). The Copper in this reaction steals electrons from Oxygen so it is responsible for oxidising oxygen, so it gets called the oxidising agent. The Oxygen gives electrons to Copper in this reaction so it is responsible for reducing copper, and therefore gets called the reducing agent in this reaction. And That's It!I hope this was useful, if you have any questions, feel free to comment or contact me.
0 Comments
What the hell is equilibrium?Equilibrium is basically when you have a reaction that goes forward and backward at the same time. An example of this in real life would be a tank of water simultaneously being filled up by tap, and emptied through a drain. For the amount of water in the tank to remain constant, the tap needs to add water at the same rate, otherwise the tank would gradually fill up or empty. Equilibrium shiftsThe position of the equilibrium is related to the amounts of substances on each side of the equilibrium reaction. For example : If there are much higher concentrations of X and Y than Z, then the equilibrium lies to the left. If there is a higher concentration of Z than X and Y then the equilibrium lies to the right. Le CHATELIER'S PrincipleLe Chatelier's Principle basically tells you which way the position of the equilibrium will move if you make changes to the equilibrium - like changing the concentrations of some of the reactants / products or change the temperature or pressure. Le chatelier's principle says that the equilibrium will shift to oppose the change you made. Changing concentrationsLets say in the equilibrium above, I removed some of substance X from the equilibrium. The equilibrium would shift left to oppose the change by making more X (and Y) from Z. The same works the other way, if I removed some of substance Z, more X and Y would react to form Z to make up for the amount you just removed. Changing TemperatureTo know which way the equilibrium shifts when you change the temperature, you need to know the ΔH for the reaction. For Example : In this example, the forward reaction is exothermic (because ΔH is negative). Le Chatelier's Principle says that the equilibrium will shift to oppose the change you've made. So if we increase the temperature, the equilibrium will shift to decrease it. To do that, it needs to use up energy. Energy is absorbed in endothermic reactions, therefore increasing the temperature favours the endothermic reaction. This of course works the other way too! If you decrease the temperature, the exothermic reaction is favoured, because it will release heat energy and change the temperature back to what it was before. Changing the pressureA quick refresher on pressure : The more particles in a fixed volume, the higher the pressure right? Le Chatelier's Principle shifts the position of the equilibrium to oppose the change you've made. If you've increased the pressure, Le Chatelier's Principle needs to reduce the number of molecules, so the equilibrium shifts towards the side with less moles of gas. In our example of X+Y⇄Z, the product side has less moles so increasing the pressure will shift the equilibrium right. I hope this was useful!So sorry I've been so inactive - I've had a hell of a lot to do! Feel free to contact me if you have any problems or questions.
Syllabus statements 3.1 - 3.8, 5.6 - 5.9, 5.13 - 5.21 Hey I've recently uploaded a new video to my YouTube channel on Organic Chemistry. But some things in this topic I thought it would be easier to understand in writing instead of in video. Read on to learn about Homologous series, catalytic Cracking and more!
What on earth are halogens?  Halogens are elements from group 7. This means they all only need 1 electron to get a full outer shell. They will try to react with other elements to get this extra electron. One important thing to remember, Halogens are diatomic, they will pair up with each other like oxygen does. Chlorine is Cl2 , Bromine Br2 etc. Click Read More to learn more about the Halogens!
What are the alkali metals?The Alkali Metals are metals from group 1 on the periodic table.
Click Read More to learn more about the Alkali Metals and how they behave!
Why does the periodic table look the way it does? Good question! Well first let's look at the periodic table and how it works. What does the periodic table show? The periodic table contains all of the elements we have ever had or created on earth. Elements after 95 (and also elements 43 and 61) are not naturally found on earth and have to be made in the lab. If you've forgotten what elements are then check out my post on them! Click Read More to learn more about how the periodic works!
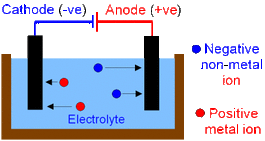
What on earth is electrolysis? Electrolysis is breaking down an ionic substance using electricity. This may sound a bit confusing so let me explain it all. If you want to learn more about electrolysis in molten salts and solutions, and their half equations read on!
Syllabus statement 1.7 and 1.8 Last post I was talking about elements, compounds and mixtures and how there is a whole big chapter on separating mixtures. Well here's the post on separating mixtures. There are a bunch of different mixtures you could have so I'll separate it up for you. If you have a solid and a solid: Is one solid magnetic? It can be removed by a magnet Does one dissolve in water? Add water, remove the insoluble solid and evaporate the water to get the soluble solid. Does one float and one sink? You can scoop the solid that floats off the top and filter the solid that sinks. If you have a liquid and a liquid: Does one have a lower boiling point than the other? You can use distillation to remove the more volatile liquid.
If there is ever a solid and liquid mixture then you only need to filter it. A simple way to find out what a mixture of liquids is for example in pen ink is to do chromatography. Click Read More to find out about Chromatography!
Syllabus statements 1.4 - 1.6, 1.9 - 1.10, 1.13 - 1.15 How do we know atoms even exist?Well, there are a few different ways actually, but I'll stick to the ones you need to know for Chemistry. The first one is diffusion. Scientists back in the day observed people smoking and thought why does the smoke disappear? Then they thought well for it to move there must be something pushing it (Newtons 2nd Law!) and the only reasonable culprit was atoms. This was backed up when a Mr. Brown was looking at pollen grains in water under a microscope when he realised that the pollen grains were jiggling around. He called this motion Brownian motion after himself like they all do. Then later on the genius Albert Einstein came along and made the link between Brown's jiggling pollen grains, diffusion and atoms and there it was. Since then our idea of what atoms are has changed, from the plum pudding model, to the nuclear model and finally to the current quantum mechanical model of the atom. For GCSE forget about the quantum mechanical atom and focus on the nuclear model. This is the 'right' one. |
AuthorI'm a 17 year old student who has a huge passion for science! Categories
All
|











 RSS Feed
RSS Feed
